Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (10): 1605-1610.doi: 10.3969/j.issn.2095-4344.0723
Previous Articles Next Articles
Socket shield technique for peri-implant tissue preservation in esthetics zone
- 1Key Laboratory of Oral and Maxillofacial Restoration and Reconstruction, 2Department of Prosthodontics, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2018-02-16Online:2018-04-08Published:2018-04-08 -
Contact:Liao Hong-bing, M.D., Professor, Department of Prosthodontics, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Chen Hui-hong, Master candidate, Laboratory of Oral and Maxillofacial Restoration and Reconstruction, Stomatology Hospital of Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, No. 81560190; the Natural Science Foundation of Guangxi Zhuang Autonomous Region, No. 2016GXNSFAA380289
CLC Number:
Cite this article
Chen Hui-hong, Wei Song-guan, Pang Bo, Wang Ling-fei, Qin Yuan, Xie Liu-rong, Liao Hong-bing. Socket shield technique for peri-implant tissue preservation in esthetics zone[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1605-1610.
share this article
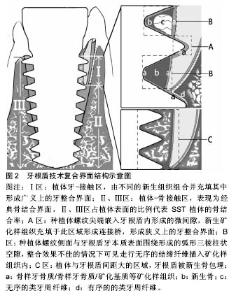
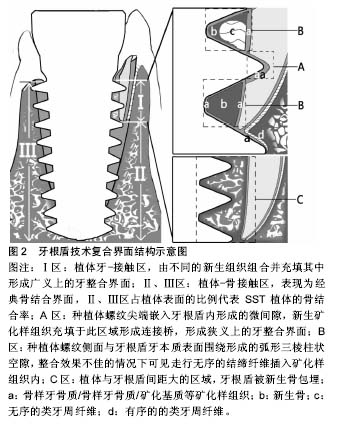
2.1 牙根盾技术理论基础 2.1.1 牙根盾技术组织保存作用 Sharpey纤维-束状骨结构是自体牙根发挥天然骨储存潜能的生理基础,通过传递功能刺激、提供生理性附着及血供支持,维持牙槽骨适应性改建的功能形态[36]。牙拔除后生理结构缺损,牙周血供遭到破坏,Sharpey纤维-束状骨在组织改建早期经历严峻的失用性萎缩及炎性破骨吸收[37-38]。由于前牙唇侧骨壁平均厚度仅1.05 mm[1,39],缺乏松质骨及骨髓腔供血,因此前牙唇侧固有牙槽束状骨及外骨板皮质骨对拔牙创伤、血供受限极为敏感,牙缺失后唇侧骨改建吸收更为显著[38]。 SST利用Sharpey纤维-束状骨的生理性骨储存潜能,保留片状牙根组织作为盾牌样结构与种植体整合,从避免拔牙创伤、提供生理性附着、维持功能刺激三个层面保存种植体周围的软硬组织,因此,作者将其翻译为牙根盾技术。牙根盾技术要求保留目的区域的自体牙根组织作为牙根盾(root shield,RS),保护相应骨壁的结构完整,屏蔽拔牙创伤引发的炎症性破骨改建[19];同时保留牙根盾的生物学宽度,从而为龈牙纤维(dentogingival fibers)、牙骨膜纤维(dentoperiosteal fibers)提供附着空间,维持游离龈、边缘骨的生理形 态[2,12,20];功能状态下牙根盾将种植体负荷通过Sharpey纤维-束状骨传递牙槽骨,为其提供更接近生理的功能刺激,避免骨组织失用性萎缩[20]。 2.1.2 牙根盾技术中的复合界面 牙根盾技术的种植体界面是由骨结合与牙整合组成的复合界面,种植体与骨壁、牙本质壁接触形成植体-骨接触区(图2Ⅱ、Ⅲ区)及植体-牙接触区(图2Ⅰ区)。骨接触区内由血液充盈并机化,实现经典骨结合界面[2,10,19];而植体-牙接触区内,种植体螺纹与牙根盾牙本质壁构成微间隙及柱状空隙,不同的新生组织相互组合并充填其中,形成牙整合界面。 新生矿化样组织分别覆盖于植体、牙根盾表面并融合成连接桥(图2aa/aba),形成类似骨结合的紧密连接(图2aa/aba)[2],Schwarz等学者将该现象定义为牙整合(dentointegration)[35],占植体-牙接触区63.5%- 67.4%[35]。在随后研究中学者根据来源及组织学特性将新生矿化样组织命名为骨样牙本质(osteodentin)[35]、骨样牙骨质(osteocementum)[2]、矿化基质(mineralized tissue matrix)[19]。骨样牙本质常见于保留有活髓的SST位点,由牙髓干细胞通过成牙本质向分化沉积而成,镜下表现为富含细胞、无明显牙本质小管结构的矿化样组织[35];骨样牙骨质由牙根盾牙周膜中的成牙骨质细胞向植体-牙接触区内迁移、分泌、矿化而成,断续状覆盖于植体粗糙面及牙本质小管表面(图2a),表现为高表达骨钙素的细胞型/非细胞型牙骨质[8,40];矿化基质(图2b)多出现于血液充盈良好的位点,由成骨细胞或成牙本质细胞分泌的细胞外基质矿化而成,其矿化程度接近骨组织,充填于植体、牙根盾之间呈现类骨粘连效果[19]。骨样牙本质、骨样牙骨质、矿化基质与牙槽骨的组织化学特性相似,组织相容性好,可沉积于种植体及牙根盾的牙本质壁表面,介导二者相互连接。 狭义上的牙整合定义与经典骨结合概念相似,指种植体与牙根盾之间有且仅有矿化样组织的结合现象,多见于植体螺纹尖端嵌入牙本质壁所形成的0.03-0.34 mm微间隙内(图2A区)[2,19,35],占植体-牙接触区63.5%- 67.4%[35]。而广义上牙整合界面包括植体-牙接触区内的结缔组织界面:植体螺纹侧面与牙根盾围成的直径0.5-1.0 mm的弧形三棱柱状空隙(dome-shaped void) (图2B区)内[10],整合效果不佳的情况下可见走行无序的结缔纤维插入矿化样组织内(图2c)[2,19,35];植体-牙接触区、骨接触区交界处牙根盾生理性牙周膜向种植体表面延伸(图2d),结构清晰、走行有序,延伸范围与种植窝的契合程度成反比[22,33-35]。 随着植体与牙根盾之间距离增宽、空隙开放程度增加,血液充盈效果改善,间隙内有新生骨长入[19],尤其在利用骨替代材料充填间隙[5,11,15,18,20]、或联合位点保存技术进行延期种植的位点中[29],牙根盾可被新生骨组织包埋(图2C),由骨结合界面取代了植体-牙接触区内牙整合界面。 2.1.3 牙根盾技术的生物力学 牙根盾的存在让SST种植体界面由经典钢性结合变为韧性-钢性复合界面,生物力学环境的改变可能对界面整体结合强度及牙槽嵴应力分布情况产生一定的影响。钢性连接包括植体-骨接触区的骨结合以及牙整合界面的牙整合/类骨结合,韧性连接界面主要包括牙整合界面的结缔组织成分、以及牙根盾的生理性牙周膜,各界面强度差异可能会削弱种植体的整体结合强度,加重骨结合界面的负担,同时牙根盾牙周膜的存在可能会引起局部力学传导模式的改变,使牙接触区与骨接触区交界处、植体颈部皮质骨成为潜在的应力集中点。既往研究表明60%以上的骨结合率(bone-implant contact,BIC)可显著降低种植体颈部骨内应力值[41],而牙根盾技术可通过控制牙根盾的保留范围获得65.3%的骨结合率[10],理论上可将植体负荷合理分散至牙槽嵴中[41],降低边缘骨应力吸收风险。但牙根盾的保留削减了植体-骨接触面积,骨结合率随牙根盾保留范围增加而降低,当骨结合率低于50%时,应力分布将集中于植体颈部的皮质骨及牙根盾内[41],造成角形吸收。因此,牙根盾范围与骨结合率、应力分布之间的关联性,以及牙整合界面强度对种植体整体长期稳定性是否存在影响,有待进一步研究考察。 2.2 牙根盾技术的临床应用 2.2.1 适应证 牙根盾技术发挥组织保存作用的前提条件是保证复合界面稳定、无炎症整合,因此,严格选择适应证、保留健康无炎症的牙根盾是成功的关键。目前公认的适应证包括:①活髓或去除牙髓炎症的无症状牙根;②无急慢性根尖周炎;③牙周附着丧失<3 mm,经系统牙周治疗后无进展性骨吸收、病理性松动[4,12,27,29]。 除了以上适应证之外,Cherel等[9,15,19,23]学者尝试对存留有固连牙、牙颈部内外吸收、慢性根尖周炎症、折裂牙根的种植位点使用牙根盾技术,并获得临床可接受的结果。对小范围慢性根尖周炎位点,Kan等[5,11,18,30]学者尝试通过搔刮、Er-YAG激光、根尖开窗手术等方式彻底去原有炎性组织,获得清洁的牙根盾技术种植窝,影像学随访发现位点根尖原有的炎性骨缺损区逐渐由小梁结构充填[12],未见种植体周围炎症表现[11-12,15]。对于存在炎症隐患的隐裂、纵裂牙位点,若裂纹未累及牙根盾设计范围,可进行常规位点预备[15];对于折裂纹累及牙根盾设计范围的位点,Baumer等[19]推荐沿裂纹预备纵向沟,将牙根盾一分为二保留,使血液充盈纵向沟,利用新生骨封闭细菌侵入通道,消除裂纹的炎症隐患,并成功应用于一例唇腭向纵裂牙,植体颈缘获得健康的龈袖口封闭,同期动物学组织切片发现纵向沟内有新生骨充满,无明显炎症细胞浸润。"

2.2.2 牙根盾技术的预备程序 牙根处理:SST位点的存留牙根需去尽腐质,并将根面降低至龈缘下1 mm[2,9],保留牙槽嵴冠方1.0-2.0 mm的生物学宽度以维持龈组织形态[9,12,16,18-20,28,30]。牙根盾的预备主要包括直接预备法及牙根分割法两种方式,直接预备法提倡“无视”牙体组织,按种植修复设计方案直接用预备钻逐级备洞至预定深度,并保留所有未松动的牙体组织作为环形牙根盾(图3c),直接预备法完全避免拔牙创伤,并可获得高度契合的种植窝。直接预备法多用于无症状的埋伏牙/根、固连牙、内外吸收牙等拔牙创伤较大的位点[9,12];分根预备法要求在根面轴角处分割牙根,拔弃无用片段后再进行预备,最大限度减少对牙根盾的挤压、调动移位,并确保完全去除根尖、精确预备根片盾,在不影响其抗折强度及组织保存效果的前提下保留最小有效范围(冠方1/3处长0-5 mm,宽相邻轴转角之间[9,11,15,18,27,29]),并将其预备为0-3 mm厚的弧形牙根盾[10-12]。常见于唇颊侧龈缘/近远中龈乳头保留位点牙根盾的预备(图3a,b)。分根预备法用于唇侧骨壁菲薄、缺损等骨吸收风险高[9,12,27,31],以及近远中间隙不足、连续种植等存在龈乳头退缩风险的前牙区种植位点[15]。 种植窝处理:种植窝的处理方式、种植时机的选择是影响SST位点整合的关键因素。牙根盾预备后需通过 搔刮、Er-YAG激光、根尖开窗手术等方式清理感染组织及碎屑,降低植体周围炎风险[5,11,18]。种植时机需根据位点组织条件及预估的植体初期稳定性来选择,若牙根盾根方有3-10 mm的可植入深度[7,10],植入扭矩达到25-45 N/cm[7,9,15],初期稳定系数(implant stability quotient,ISQ)达60-70 s时可选择即刻植入种植体[7,18];对于预估植体稳定性不佳的位点,可选择用吸收胶原填塞SST窝洞做位点保存,3-6个月后在充满新生骨的原位点进行延期种植[29]。 植体-牙间隙处理:植体-牙间隙处理涉及到距离控制及生物材料的应用,早期技术理念提倡将植体紧贴牙根盾植入以利于牙整合,并在微间隙内涂抹釉质基质蛋白,促进骨样牙骨质沉积形成牙整合界面[9,40];后期学者认为增加植体-牙间隙可改善血液充盈效果[9],联合骨替代材料、富含血小板生长因子及纤维蛋白促间隙内成骨,可增加牙根盾技术位点的植体-植骨结合率[5,11,15,18,20]。 2.3 SST位点种植体预后 2.3.1 临床评价 牙根盾技术尚无公认的成功标准,作者参考1986年瑞典Albrektsson口腔种植成功标准及Davarpanah牙根盾评估指标[27。42],整理提出牙根盾技术位点的评价标准:①种植体无动度;②种植体无持续或不可逆的症状,如疼痛、感觉异常等;③牙根盾不穿通龈袖口,或穿通后龈袖口无炎症;④X-ray显示植体-骨界面、植体-牙界面无透射区;牙根盾周围无异常透射影,无进行性再吸收,或牙根盾吸收区由骨阻射影充填;⑤种植体功能负重第1年,垂直骨吸收量不超过不超过1.5 mm,往后每年吸收量小于0.2 mm。 临床对照试验报道了10例牙根盾技术和16例拔牙即刻种植患者,在2年随访期间,2组种植体均无松动,成功率100%,牙根盾技术组唇侧骨高度共降低0.8 mm、软组织体积退缩2%,拔牙即刻种植组骨高度降低5 mm、软组织体积退缩18%,2组种植位点均获得协调的龈曲线,试验结果表明牙根盾技术组织保存效果与常规即刻种植技术有可比性[5]。27篇病例报告中,最长随访时间为9年[21],共出现5例并发症:Davarpanah等[27]将牙根盾技术应用于无症状水平埋伏牙位点,并报告1例(1/7) 8.5 mm短植体因植入深度不足术后4个月松动脱落,存留率为86%;Lagas等[31]学者报道1例(1/16)牙根盾周围炎,检查发现植体稳定性未受影响,手术刮除牙根组织后保留植体原位愈合;Schwarz等[12,21,27]报道的3例牙根盾无菌性吸收现象,种植体功能负重均未受影响,其中1例种植体颈部牙根组织完全置换性吸收后,牙槽骨高度稳定于植体第二、三螺纹之间,未出现不可控制的再吸收现象[21]。 2.3.2 牙根盾技术并发症 牙根盾、种植体周围炎:在经典骨结合理念中,种植窝内残留的牙体、结缔组织被视为影响植体整合的隐患[42],机械预备对残留炎性组织的激惹是引起SST位点炎性并发症的主要诱因。牙根盾的受压、调动将增加Sharpey’s纤维-束状骨复合体的易感性,若原有炎性组织未得到彻底清理,受激惹后炎症将沿着牙周间隙扩散,引起牙根盾周围炎。并且受压移位的牙根盾存在病理性动度,影响位点整合,并成为连接植窝与残留的炎性组织之间的潜在通道,引起植体周围炎症性快速骨丧失导致种植失败[24]。因此,SST位点预备过程中彻底去除牙根根尖、清理种植窝内炎症组织、避免牙根盾过度调动移位,是降低炎性并发症的关键。 种植体松动脱落:SST位点种植体的长期稳定性主要由复合界面中骨结合提供,若牙根盾保留范围过大、种植体植入深度不足将削减植体-骨结合面积,过低的骨结合率无法承受功能负荷导致种植体松动脱落。因此原则上要求,在不影响组织保存效果的前提下应尽量减小牙根盾面积,充分利用可植入深度[7,10],以增加植体-骨接触区,确保42.4%-65.3%的骨结合率[10]。 牙根盾无菌性吸收:Schwarz等[12,21,23]报道的3例牙根盾吸收位点,并推测植体过负荷、牙根固连为牙根盾无菌性吸收的诱因。在过负荷49个月的牙根盾技术位点中,牙根盾冠方的牙本质出现与牙槽骨同等程度的角形吸收,表现为典型过负荷植体周组织适应性改建[27];存在骨黏连的牙根盾易出现无菌性置换吸收,5年影像学随访显示种植体颈部的固连牙根完全置换性吸收后,牙槽骨高度稳定于植体第二、三螺纹之间,植体周围无异常透射影[21],并且牙根盾吸收区及原牙根炎症骨缺损区均由骨密度影像充填[12]。然而牙根盾吸收现象是否会对种植体存留率及组织保存效果造成影响、吸收区是否会被除了骨以外的组织替代进而成为炎症隐患,有待进一步考查。 "

| [1]Masaki C, Nakamoto T, Mukaibo T, et al. Strategies for alveolar ridge reconstruction and preservation for implant therapy. J Prosthodont Res. 2015;59(4):220-228. [2]Hürzeler MB, Zuhr O, Schupbach P, et al. The socket-shield technique: a proof-of-principle report. J Clin Periodontol. 2010;37(9):855-862. [3]Gharpure AS, Bhatavadekar NB. Current Evidence on the Socket-Shield Technique: A Systematic Review. J Oral Implantol. 2017;43(5):395-403. [4]Khalil J. Socket shield and immediate implantation. Porto, Portugal: Universidade Fernando Pessoa, 2016.[5]Bramanti E, Norcia A, Cicciù M, et al. Postextraction Dental Implant in the Aesthetic Zone, Socket Shield Technique Versus Conventional Protocol. J Craniofac Surg. 2018. doi: 10.1097/SCS.0000000000004419. [6]Szmukler-Moncler S, Davarpanah M. Reliability of the root-implant interface in unconventionally placed implants: An up to 6 year follow-up of 23 implants covering 3 distinct clinical applications. Clin Oral Implants Res. 2009.[7]Troiano M, Benincasal M, Sánchez P. Bundle bone preservation with Root-T-Belt: Case study. Ann Oral Maxillofac Surg. 2014;2(1):1-7.[8]Guarnieri R, Giardino L, Crespi R, et al. Cementum formation around a titanium implant: a case report. Int J Oral Maxillofac Implants. 2002;17(5):729-732.[9]Saeidi Pour R, Zuhr O, Hürzeler M, et al. Clinical Benefits of the Immediate Implant Socket Shield Technique. J Esthet Restor Dent. 2017;29(2):93-101.[10]Guirado JL, Troiano M, López-López PJ, et al. Different configuration of socket shield technique in peri-implant bone preservation: An experimental study in dog mandible. Ann Anat. 2016;208:109-115.[11]Gluckman H, Du Toit J, Salama M. Guided bone regeneration of a fenestration complication atimmediate implant placement simultaneous to the socketshieldtechnique. Int Dent Afr Ed. 2014;5(4):58-66.[12]Siormpas KD, Mitsias ME, Kontsiotou-Siormpa E, et al. Immediate implant placement in the esthetic zone utilizing the "root-membrane" technique: clinical results up to 5 years postloading. Int J Oral Maxillofac Implants. 2014;29(6): 1397-1405. [13]Holbrook SE. Model-Guided Flapless Immediate Implant Placement and Provisionalization in the Esthetic Zone Utilizing a Nanostructured Titanium Implant: A Case Report. J Oral Implantol. 2016;42(1):98-103. [14]Barbosa FT, Carmona D, Fal V, et al. A novel approach for a volumetric variation assessment after immediate implants using Socket Shield Technique: A report of a serie of cases. Europerio. 2015.[15]Cherel F, Etienne D. Papilla preservation between two implants: a modified socket-shield technique to maintain the scalloped anatomy? A case report. Quintessence Int. 2014; 45(1):23-30. [16]Mahajan T, Massey N, Bajwa W. Socket shield technique. Indian Dendtal J. 2015;7:31-34.[17]Al-Dary HH. The socket shield technique: a case report. Smile Dental J. 2013;8(1):32-36.[18]Gluckman H, Du Toit J, Salama M. The socket-shield technique to support the buccofacialtissues at immediate implant placement. Int Dent Afr Ed. 2015;5(3):6-14.[19]Bäumer D, Zuhr O, Rebele S, et al. The socket-shield technique: first histological, clinical, and volumetrical observations after separation of the buccal tooth segment – a pilot study. Clin Implant Dent Relat Res. 2015;17(1):71-82. [20]Gluckman H, Du Toit J, Salama M. The Pontic-Shield: Partial Extraction Therapy for Ridge Preservation and Pontic Site Development. Int J Periodontics Restorative Dent. 2016; 36(3):417-423.[21]Szmukler-Moncler S, Davarpanah M, Davarpanah K, et al. Unconventional Implant Placement Part III: Implant Placement Encroaching upon Residual Roots - A Report of Six Cases. Clin Implant Dent Relat Res. 2015;17 Suppl 2: e396-405. [22]Davarpahah SM. Unconventional implant treatment. II. Implant placed through impacted teeth: 3 case reports. Int J Periodontics Restorative Dent. 2009;29:405-413.[23]Davarpanah M, Szmukler-Moncler S. Unconventional implant treatment: I. Implant placement in contact with ankylosed root fragments. A series of five case reports. Clin Oral Implants Res.2009;20(8):851-856. [24]Langer L, Langer B, Salem D. Unintentional root fragment retention in proximity to dental implants: a series of six human case reports. Int J Periodontics Restorative Dent. 2015;35(3): 305-313. [25]Baumer D, Zuhr O, Hurzeler M. “Socket Shield” – Innovations for immediate implant placement in the esthetic zone. Implantologie. 2015;23(4):453-461.[26]Kuit H. Socket shield-technique: 3 yearr follow-up. Tandartspraktijk. 2014;35(8):4-11.[27]Davarpanah M, Szmukler-Moncler S, Davarpanah K, et al. Unconventional transradicular implant placement to avoid invasive surgeries: toward a potential paradigm shift. Rev Stomatol Chir Maxillofac. 2012;113(4):335-349. [28]Chen C, Pan Y. Socket shield technique for ridge preservation a case report. Prosthondontics Implantology. 2013;2:16-21.[29]Glocker M, Attin T, Schmidlin P. Ridge preservation with modified “socket-shield” technique a methodological case series. Dent J. 2014;2:11-21.[30]Kan JY, Rungcharassaeng K. Proximal socket shield for interimplant papilla preservation in the esthetic zone. Int J Periodontics Restorative Dent. 2013;33(1):e24-31.[31]Lagas LJ, Pepplinkhuizen JJ, Bergé SJ, et al. Implant placement in the aesthetic zone: the socket-shield-technique. Ned Tijdschr Tandheelkd. 2015;122(1):33-36. [32]Buser D, Warrer K, Karring T. Formation of a periodontal ligament around titanium implants. J Periodontol. 1990;61(9): 597-601.[33]Gray JL, Vernino AR. The interface between retained roots and dental implants: a histologic study in baboons. J Periodontol. 2004;75(8):1102-1106.[34]Warrer K, Karring T, Gotfredsen K. Periodontal ligament formation around different types of dental titanium implants. I. The self-tapping screw type implant system. J Periodontol. 1993;64(1):29-34.[35]Schwarz F, Mihatovic I, Golubovic V, et al. Dentointegration of a titanium implant: a case report. Oral Maxillofac Surg. 2013; 17(3):235-241. [36]Dugan DJ, Getz JB, Epker BN. Root banking to preserve alveolar bone: a review and clinical recommendation. J Am Dent Assoc. 1981;103(5):737-743.[37]Cardaropoli G, Araújo M, Lindhe J. Dynamics of bone tissue formation in tooth extraction sites. An experimental study in dogs. J Clin Periodontol. 2003;30(9):809-818.[38]Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.[39]Nowzari H, Molayem S, Chiu CH, et al. Cone beam computed tomographic measurement of maxillary central incisors to determine prevalence of facial alveolar bone width ≥2 mm. Clin Implant Dent Relat Res. 2012;14(4):595-602. [40]Hürzeler MB, Zuhr O, Schupbach P, et al. The socket-shield technique: a proof-of-principle report. J Clin Periodontol. 2010;37(9):855-862. [41]Petrie CS, Williams JL. Comparative evaluation of implant designs: influence of diameter, length, and taper on strains in the alveolar crest. A three-dimensional finite-element analysis. Clin Oral Implants Res. 2005;16(4):486-494.[42]Albrektsson T, Zarb G, Worthington P, et al. The long-term efficacy of currently used dental implants: a review and proposed criteria of success. Int J Oral Maxillofac Implants. 1986;1(1):11-25.[43]Brånemark PI, Hansson BO, Adell R, et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl. 1977;16:1-132. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||